Use of a mixture of coal and oil as an additive for selective reduction of lateritic ore by the Caron process Technical paper
Main Article Content
Abstract
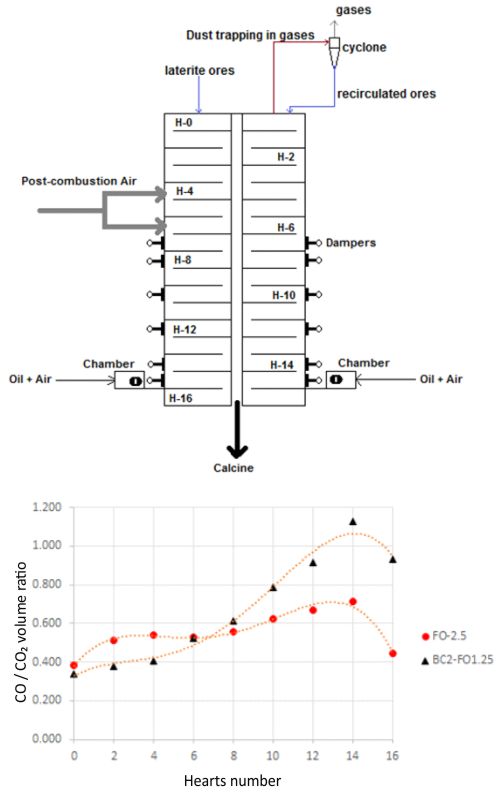
Lateritic ores constitute the main source of raw material for extraction of Ni and Co by the Caron process. Consumption of oil in the reduction furnace is one of the key indicators if the metallurgical process is economical. To date it has not been possible to replace the additive fuel oil that is used at commercial scales, therefore, the aim of this study was to partially replace the oil with bituminous coal on a pilot scale by using a mixture of 2 % coal and 1.25 % oil as the reducer additive. Phases of the reduced/leached ores were analyzed by powder X-ray diffraction, while the metallic state of the ore was determined by leaching the reacted samples with a bromine-ethanol solution followed by the atomic absorption spectrometry analysis. Extractions of Ni and Co were confirmed by leaching the reduced ore with ammoniacal-ammonium carbonate solutions. It was observed that the mixture used as a reducer additive can replace the fuel oil since it allows the adequate transformation of the main mineralogical phases of the laterite ore during the reduction process and the average extraction yields of Ni and Co for ~3 and ~8 %, respectively. Although the effect of bituminous coal particle size in the process was not analyzed, the reducing mixture ensured that the Caron process was more efficient.
Article Details
Issue
Section

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors who publish with this journal agree to the following terms:
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
Authors grant to the Publisher the following rights to the manuscript, including any supplemental material, and any parts, extracts or elements thereof:
- the right to reproduce and distribute the Manuscript in printed form, including print-on-demand;
- the right to produce prepublications, reprints, and special editions of the Manuscript;
- the right to translate the Manuscript into other languages;
- the right to reproduce the Manuscript using photomechanical or similar means including, but not limited to photocopy, and the right to distribute these reproductions;
- the right to reproduce and distribute the Manuscript electronically or optically on any and all data carriers or storage media – especially in machine readable/digitalized form on data carriers such as hard drive, CD-Rom, DVD, Blu-ray Disc (BD), Mini-Disk, data tape – and the right to reproduce and distribute the Article via these data carriers;
- the right to store the Manuscript in databases, including online databases, and the right of transmission of the Manuscript in all technical systems and modes;
- the right to make the Manuscript available to the public or to closed user groups on individual demand, for use on monitors or other readers (including e-books), and in printable form for the user, either via the internet, other online services, or via internal or external networks.
How to Cite
References
Bartzas G, Tsakiridis PE, Komnitsas K. Nickel industry: Heavy metal(loid)s contamination - sources, environmental impacts and recent advances on waste valorization. Curr Opin Environ Sci Heal. 2021; 21: 100253 https://doi.org/10.1016/j.coesh.2021.100253
Mitterecker J, Košević M, Stopic S, Friedrich B, Panić V, Stevanović J, Mihailović M. Electrochemical Investigation of Lateritic Ore Leaching Solutions for Ni and Co Ions Extraction. Metals (Basel). 2022; 12(2): 325 https://doi.org/10.3390/met12020325
Al-Khirbash SA. Mineralogical characterization of low-grade nickel laterites from the North Oman Mountains: Using mineral liberation analyses - scanning electron microscopy-based automated quantitative mineralogy. Ore Geol Rev. 2020; 120: 103429 https://doi.org/10.1016/j.oregeorev.2020.103429
Gleeson SA, Butt CRM, Elias M. Nickel Laterites. SEG Discov 2003; (54): 1-18 https://doi.org/10.5382/SEGnews.2003-54.fea.
Domenech C, Galí S, Villanova-de-Benavent C, Soler J, J P. Reactive transport model of the formation of oxide type Ni-laterite profiles (Punta Gorda, Moa Bay, Cuba. Mineralium Deposita. 2017; 52(7): 993-1010
Aiglsperger T, Proenza JA, Lewis JF, et al. Critical metals (REE, Sc, PGE) in Ni laterites from Cuba and the Dominican Republic. Ore Geol Rev. 2016; 73: 127-147. https://doi.org/10.1016/j.oregeorev.2015.10.010
Oxley A, Barcza N. Hydro-pyro integration in the processing of nickel laterites. Miner Eng. 2013; 54: 2-13. https://doi.org/10.1016/j.mineng.2013.02.012.
Caron MH. “Fundamental and practical factors in ammonia leaching of nickel and cobalt ores” JOM-Journal of the Minerals, Metals and Materials Society, 1950; 2(1): 67-90. https://doi.org/10.1007/BF03398981
Caron MH. “Separation of Nickel and Cobalt” JOM-Journal of the Minerals, Metals and Materials Society, 1950; 2(1): 91-103. https://doi.org/10.1007/BF03398982
Kießling F, Stopic S, Gürmen S, Friedrich B. Recovery of Diamond and Cobalt Powders from Polycrystalline Drawing Die Blanks via Ultrasound Assisted Leaching Process—Part 2: Kinetics and Mechanisms. Metals (Basel), 2020; 10(6): 741. https://doi.org/10.3390/met10060741
Palma HJA, Legrá AL, Urgellés AL, Gálvez E, Castillo J. Post-combustion Effect on Nickel and Cobalt Extractions from the Caron Process. In: Proceedings of Fourth International Conference on Inventive Material Science Applications, ICIMA 2021. Coimbatore, India, 2022, pp. 515-527. https://doi.org/10.1007/978-981-16-4321-7_43
Rodriguez R. Reduction in energy cost in Cuban Caron Process Plants. In: International Laterite Nickel Symposium 2004 (as held during the 2004 TMS Annual Meeting). The Minerals, Metals & Materials Society 2004, pp. 657-664. ISBN: 0-87339-550-6.
Canterford Jh. Oxide Ores of Nickel — The Australian Situation. Miner Process Extr Metall Rev. 1983; 1(1-2): 85-109. https://doi.org/10.1080/08827508308952590
Shofi A, Supriyatna YI, Prasetyo AB. Selective Reduction of Southeast Sulawesi Nickel Laterite using Palm Kernel Shell Charcoal: Kinetic Studies with Addition of Na2SO4 and NaCl as Additives. Bull Chem React Eng Catal. 2020; 15(2): 501-513. https://doi.org/10.9767/bcrec.15.2.7733.501-513
Coello-Velázquez AL, Quijano Arteaga V, Menéndez-Aguado JM, Pole FM, Llorente L. Use of the Swebrec Function to Model Particle Size Distribution in an Industrial-Scale Ni-Co Ore Grinding Circuit. Metals (Basel). 2019; 9(8): 882. https://doi.org/10.3390/met9080882
De Graaf JE. The treatment of lateritic nickel ores — a further study of the caron process and other possible improvements. Part I. Effect of reduction conditions. Hydrometallurgy. 1979; 5(1): 47-65. https://doi.org/10.1016/0304-386X(79)90027-6
Pickles CA, Elliott R. Thermodynamic analysis of selective reduction of nickeliferous limonitic laterite ore by carbon monoxide. Miner Process Extr Metal.l 2015; 124(4): 208-216. https://doi.org/10.1179/1743285515Y.0000000009
Pickles CA, Anthony W. A Thermodynamic Study of the Reduction of a Limonitic Laterite Ore by Methane. High Temp Mater Process. 2018; 37(9-10): 909-19. https://doi.org/10.1515/htmp-2017-0106
Pickles CA, Anthony W. Thermodynamic modelling of the reduction of a saprolitic laterite ore by methane. Miner Eng. 2018; 120: 47-59. https://doi.org/10.1016/j.mineng.2018.02.006
de Alvarenga Oliveira V, dos Santos CG, de Albuquerque Brocchi E. Assessing the Influence of NaCl on the Reduction of a Siliceous Laterite Nickel Ore Under Caron Process Conditions. Metall Mater Trans B. 2019; 50(3): 1309-1321. https://doi.org/10.1007/s11663-019-01552-w
Valix M, Cheung W. Effect of sulfur on the mineral phases of laterite ores at high temperature reduction. Miner Eng. 2002; 15(7): 523-530. https://doi.org/10.1016/S0892-6875(02)00069-9
Ilyas S, Srivastava RR, Kim H, Ilyas N, Sattar R. Extraction of nickel and cobalt from a laterite ore using the carbothermic reduction roasting-ammoniacal leaching process. Sep Purif Technol. 2020; 232: 115971. https://doi.org/10.1016/j.seppur.2019.115971
Angulo-Palma HJ, Legrá-Legrá A, Hernández-Pedrera C, Lamorú-Urgellés A, Vega-Cala RJ. Efecto de la sustitución del petróleo aditivo por carbón bituminoso en el proceso de reducción de lateritas. Eff Substit Addit Oil with Bitum Coal Process Reducing Laterites. 2018; 38(3): 750-764. (Spanish)
Angulo-palma HJ, Legr A, Hern C, Lamor A, Toro-villarroel N. Reducción de menas lateríticas utilizando como aditivo mezclas de carbón bituminoso y petróleo. Tecnol Química. 2020; 40(1): 93-105. (Spanish)
Angulo-palma HJ, Terencio-guevara PL, Legrá-Legrá A, Videaux-arcia L. Análisis especiales en un horno de reducción de níquel a escala de Planta Piloto Special Analysis in a Nickel Reduction Furnace at Pilot Plant scale. Tecnol Química. 2017; 37(3): 484-499. (Spanish)
Basulto L. Evaluación de la sustitución de fuel-oil por finos de carbón antracita como aditivo en la empresa Comandante Ernesto Che Guevara 2018. http://ninive.ismm.edu.cu/handle/123456789/1706 (Spanish)
Ilyas S, Kim H, Srivastava RR. Carbothermic Reduction Roasting of a Low-Grade Nickel Laterite Ore in the Modified Caron Process. In: Ni-Co 2021 5th Int Symp Nickel Cobalt 2021: 317-328. https://doi.org/10.1007/978-3-030-65647-8_27
Suarez JC, Villanueva GAA, Fuxa AMDLC, González PM, Urtafe IL, Laurencio OP, Rodríguez ME. Hydrometallurgical process for the recovery of nickel and cobalt by ammoniacal leaching. US 6,524,367 B1, 2003
Angulo-Palma HJ, Legrá-Legrá A, Hernández-Pedrera C. Modelos para determinar variables importantes en el proceso Caron. Parte I: Consumo específico de energía en molienda. Opuntia Brava. 2021; 13: 149-57. (Spanish)
Sant B. Determination of metallic iron, iron(II) oxide, and iron(III) oxide in a mixture. Talanta. 1968; 15(12): 1483-1486. https://doi.org/10.1016/0039-9140(68)80211-5
Chander S, Sharma VN. Reduction roasting/ammonia leaching of nickeliferous laterites. Hydrometallurgy. 1981; 7(4): 315-327. https://doi.org/10.1016/0304-386X(81)90029-3
Valix M, Cheung WH. Study of phase transformation of laterite ores at high temperature. Miner Eng. 2002; 15(8): 607-612. https://doi.org/10.1016/S0892-6875(02)00068-7
O’Connor F, Cheung WH, Valix M. Reduction roasting of limonite ores: effect of dehydroxylation. Int J Miner Process. 2006; 80(2-4): 88-99 https://doi.org/10.1016/j.minpro.2004.05.003
Pintowantoro S, Widyartha AB, Setiyorini Y, Abdul F. Sodium Thiosulfate and Natural Sulfur: Novel Potential Additives for Selective Reduction of Limonitic Laterite Ore. J Sustain Metall. .2021; 7(2): 481-494. https://doi.org/10.1007/s40831-021-00352-4
Cabrera G, Gómez JM, Hernández I, Coto O, Cantero D. Different strategies for recovering metals from CARON process residue. J Hazard Mater. 2011; 189(3): 836-842. https://doi.org/10.1016/j.jhazmat.2011.03.048
Rojas-Purón AL, Turro-Breffe A. Composición mineralógica de las colas del proceso Caron en Moa, Holguín, Cuba. Minería y Geol. 2003; 19:8. (Spanish)
Miranda J, Chaviano L, Miranda J. Nuevas interpretaciones quimico-mineralogicas de las menas lateriticas y serpentinicas a traves del proceso pirometalurgico en la tecnologia carbonato-amoniacal. Rev Cuba Química. 2002; 14: 42-51. (Spanish)
Sánchez-Ramos S, Doménech-Carbó A, Gimeno-Adelantado JV, Peris-Vicente J, Valle-Algarra FM. Thermal decomposition of chromite spinel with chlorite admixture. Thermochim Acta. 2008; 476(1-2): 11-19. https://doi.org/10.1016/j.tca.2008.07.003
Chang-Cardona AR, Rojas-Vargas A. Comportamiento electroquímico del Fe y sus iones durante la lixiviación estándar QT de minerales reducidos en el proceso “Caron” con diferentes aireaciones. Tecnol Química. 2014; 34: 117-127. (Spanish)
Rojas-Vargas A, Sánchez-Guillen C, Magaña-Haynes ME, Hernández-Pedrera C. Extracción potencial de níquel y cobalto con mineral laterítico de mina “Pinares de Mayarí” en la tecnología Caron. Parte I. Tecnol Química. 2021; 41: 519531. (Spanish)
[Rojas Vargas A, Magaña Haynes ME, Riverón AR. Lixiviación carbonato amoniacal: estimación del níquel disuelto en el efluente de destilación. Rev Metal. 2019; 55(3): 149.https://doi.org/10.3989/revmetalm.149. (Spanish)